
Matt Gibbs, president of Capital Rx, to discusses alternative career paths for students, including independently owned PBMs.

Matt Gibbs, president of Capital Rx, to discusses alternative career paths for students, including independently owned PBMs.
Antibodies help neutralize the virus, though the levels decrease at the 6-month mark, data indicate.

Dental opioid prescriptions are associated with risk of addiction and overdose, but most states are enacting policies that limit the duration of prescriptions for acute pain.
Educating patients, posting on social media, and countering misinformation are critical strategies.
Deucravacitinib is an oral, selective, allosteric tyrosine kinase 2 inhibitor that inhibits cytokine signaling in psoriasis pathogenesis.

November 27, 2023, marks date set by statute for full implementation of Drug Supply Chain Security Act.

Key trends such as consolidation create an opportunity for independent pharmacies with a community-based approach.
Data from a randomized clinical trial supports previous clinical findings on SGLT2 inhibitors and their ability to slow the progression of chronic kidney disease.

This week's episode features Amina Abubakar, PharmD, AAHIVP, CEO of Avant Pharmacy & Wellness Center and president of Avant Institute, who discusses her experience in the pharmacy industry and the communities she serves.

There is a lack of funding for attention-deficit hyperactivity disorder and coinciding psychological research compared to the funds generated for autism research.
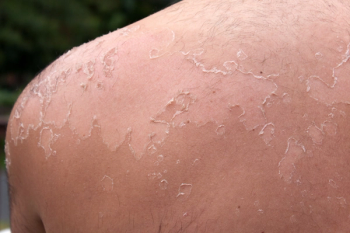
Soft tissue reconstruction solutions can leverage a patient’s natural healing response.
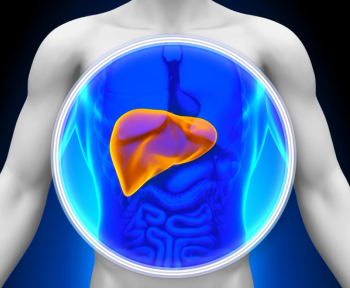
Individuals with higher caffeine levels were less likely to have liver fibrosis, while higher levels of non-caffeine coffee components were associated with reduced fatty liver index scores.

Products range from natural eye relief to sleep aid.
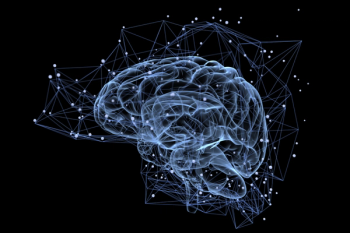
The model can predict the chronological ages of cognitively normal participants with an average absolute error of 2.3 years, which is about 1 year more accurate than an existing model.

Federal court weighs whether termination that followed was aged-based discrimination under state's Human Rights Act.

An app using AI to evaluate blood sugar data, concerning trends, and recommend management tools will be a part of a clinical trial in 2024 for further assessment.

The vaccine was 88% effective at preventing pertussis among infants younger than 2 months when administered during the third trimester of pregnancy.

COVID-19 infection was significantly associated with increased risks of several conditions, including loss of smell and taste, concentration, and memory impairment.
Energy crisis in Europe puts strain on production and could have ramifications that extend beyond the continent's borders.
It is too early to know how patients could potentially change their microbiome to reduce their diabetes risk.

Study notes that evaluating changes in the proportion of hospitalizations for mental health conditions among adolescents is vital to ensure the prioritization of public health programs that address psychiatric health care needs that emerged during the COVID-19 pandemic.

The scholarship is accepting applications until February 28, 2023, and 4 pharmacy school students will be announced in April.

Clonidine works on adrenergic receptors in the brain, which are known for their role in fight-or-flight responses and are believed to be activated in post-traumatic stress disorder.

Label update removes a previous limitation that stated the medication should not be used as initial therapy for treating patients with type 2 diabetes.

Ronna Hauser of the National Community Pharmacists Association and Olin and Scotty Sykes, accountants from Sykes & Company, discuss DIR changes.

Analysis focuses on why some individuals with a genetic predisposition develop AMD, while others are spared.

Study is the first to examine calciferol in brain tissue, which may help investigators understand dementia and its causes.

Providers use "Kitchen Sink Approach" because cure proves elusive, symptomatic treatment is sketchy.

Counseling sessions offer opportunity to educate about appropriate dosing, potential adverse effects.

Analysis focuses on causal effects while accounting for other environmental and genetic factors, such as a family history and socioeconomic disadvantages.