
Students may be hesitant to discuss migraines, which can lead to absences, difficulties in personal life, financial burdens, and poor mental health.

Students may be hesitant to discuss migraines, which can lead to absences, difficulties in personal life, financial burdens, and poor mental health.

Analysis shows average monthly premiums for stand-alone plans will rise 10% to $43.
Relative efficacy of nirmatrelvir–ritonavir appears similar across risk groups, but older adults at risk of severe COVID-19 may benefit more.

Study findings confirmed that in asthma, chronic obstructive pulmonary disease, HIV, and influenza, census data differed from the epidemiological data.

The new findings, which include updated data, contradict results of a previous analysis by the same investigators.

Amanda Ryan, the director of Education for the Pharmacy Quality Alliance (PQA), discusses a new PQA program to teach pharmacy and other health professions students the basics of quality measurement and strategies to improve medication safety, adherence and appropriate use.
Study shows no robust differences in blood pressure, heart rate, lipids, and glucose measurements in children conceived using assisted reproductive technologies.
Results of 2 phase 3 clinical trials demonstrate better response rate beyond the primary endpoint analysis at week 16 to more than 55 at week 55.

Investigators focus on whether legalization of the drug is associated with an increase in health care use for the psychotic disorder.
Streptolysin O appears to influence the pathogenesis of Streptococcus group A and direct mutation-mediated virulence in a host.

The association with subgroups of gene variations based on expression or function should be assessed in larger studies, investigators say.

Expanded indication for abrocitinib (Cibinqo; Pfizer Inc) includes patients 12 to <18 years of age with refractory, moderate-to-severe atopic dermatitis that is not adequately controlled with other systemic medications.
Learn strategies to safeguard use of the intravenous administration products to protect patients.

Despite the large number of prescription products available, nondrug therapies often work just as well.

American Heart Association maintains stance on preferably obtaining essential nutrients through dietary means.
A booster vaccine for COVID-19 increased the concentration of antibodies in older patients more than younger patients.
Several studies have noted a potential link between methotrexate and an increased risk of skin cancer.

Pharmacists play a vital role in educating patients about differentiating among the common cold, COVID-19, influenza, and respiratory syncytial virus.

A total of 150 respondents rated their satisfaction with total annual compensation as 4.15 on a scale of 1 to 7, down from 4.41 in 2021.
Prior to the development of vaccines and direct-acting antivirals, hepatitis infections were treated through methods of prevention.

Long COVID can impact quality of life, with some symptoms lasting for 1 year before the patient recovers to baseline.
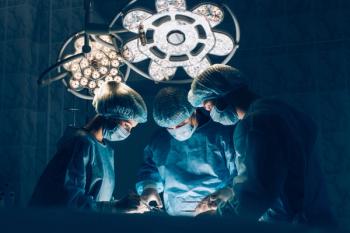
Physician outlines the process developed at The Ohio State University Comprehensive Cancer Center to help manage patient pain before, during, and after surgery without the use of opioids.
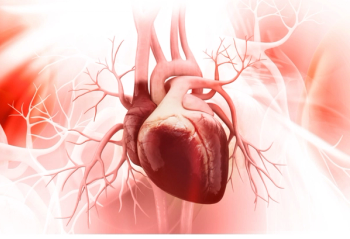
Despite medical advances in the past few decades, rates of PCAD in patients with diabetes, hypertension, obesity continue to rise.

Multiple adverse outcomes were associated with an even higher risk of heart disease.

Aflibercept injection is also indicted for the treatment of neovascular age-related macular degeneration, macular edema following retinal vein occlusion, diabetic macular edema, and diabetic retinopathy.

Individuals with more fast-food options in their communities had 13% higher odds of incident stroke.
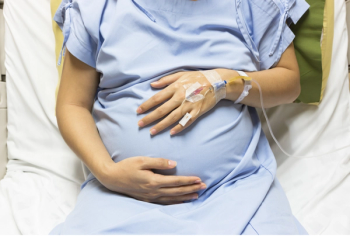
JNJ-80202135 (nipocalimab) treats hemolytic disease of the fetus and newborn caused by maternal red blood cells.

In a fact sheet released by the White House, President Joseph Biden called a $35 monthly cap on insulin for all Americans a “commonsense, life-saving protection.”

Vitamin D supplementation is a vital and inexpensive way to prevent and treat deficiency; however, its use in preventing and treating other conditions is less clear.

On February 6, 2023, Pharmacy Times also learned that the FDA just accepted a new drug application (NDA) for this treatment.