Four new subtypes of cells within triple-negative breast cancer have been uncovered, containing promising new therapeutic targets for the disease.

Four new subtypes of cells within triple-negative breast cancer have been uncovered, containing promising new therapeutic targets for the disease.

More than 10 million adults over age 50 have osteoporosis.
Flu vaccination will be particularly important in the 2020-2021 season to lessen the burden on the health care system and ensure adequate resources amid the coronavirus disease 2019 pandemic.
Despite, antiretroviral therapy, HIV can hide in blood and tissue.

Continuous glucose monitoring has revolutionized the way patients with diabetes can monitor their glucose.
SalivaDirect, developed by researchers at the Yale School of Public Health, is currently in use by the National Basketball Association to test players and staff for COVID-19.
Fingolimod (Gilenya), which is approved by the FDA to treat multiple sclerosis flare-ups, may also block HIV and reduce the latent reservoir.

Now, more than ever, is the time that nurses, pharmacists, pharmacy technicians, and physicians can help those who need access to life-saving medicines, and Dispensary of Hope is a resource.

As misconceptions surround some OTC supplements, changes to the diet may be beneficial in supporting brain health in populations experiencing age-related memory loss.

A patient has unexplained symptoms that appear to be severe anxiety.

Elderly patients and infants with dysphasia who cannot or will not swallow solid medications can take liquid or sprinkle ones.

Cancer survivors’ lean body mass found to be lower than the general population, which was connected with prior abdominal or pelvic radiation.

Stephanie Moss, PharmD, MS, is going live each Wednesday evening on the Pharmacy Times Instagram and Facebook pages. Tune in each week to learn more about establishing a presence on social media and join the discussion.

An expert panel discussion for clinicians on the most important factors surrounding a safe return-to-school plan, including testing, masking, social distancing, vaccine prioritization and more.

Unique model demonstrates how clinical pharmacy services could be integrated into an interprofessional telehealth clinic.
Researchers attempt to address a lack of evidence showing that schools have a role in the transmission of COVID-19.
Top news of the week from Pharmacy Times®.

What constipation remedy did Lewis and Clark take on their famed expedition?
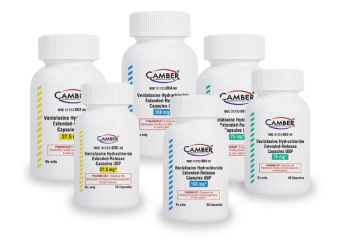
Venlafaxine Hydrochloride ER capsules is used to treat major depressive disorder, anxiety and panic disorder.

Leaders of Operation Warp Speed optimistic for a coronavirus disease 2019 vaccine following multi-billion dollar investments in several candidates.
NACDS states that although prescription drug discount cards were put in place to reduce out-of-pocket costs, states have been using the data in a manner inconsistent with federal law and policy.

Stephanie Young Moss, PharmD, MS, discussed how to begin finding your niche as a pharmacist on social media.

Vitolarsen is the second FDA-approved targeted treatment for patients with Duchenne muscular dystrophy who have a confirmed mutation of the gene that is amenable to exon 53 skipping.

Analysis suggests that the current diabetes screening criteria, despite covering about a quarter of US children and adolescents, do not capture many youth with hyperglycemia.
Like so many other conferences and events, ASCO transitioned to an online format because of the coronavirus disease 2019 (COVID-19) outbreak.

The 2020 NCPA Annual Convention will be held exclusively online October 18-19, 2020.
Study shows significant genomic differences between tumors caused by different strains of human papillomavirus.
The peptide, also known as Pept-ins, already proved to be a valuable approach to tackle bacterial pathogens or slow down tumor growth, according to the study authors.

Symptoms of polycystic ovary syndrome, such as obesity and diabetes, can lead to a 19% higher risk of heart disease.

BAM15 may help reduce the amount of food a person eats or the calories their bodies absorb.