Six months following discharge from the hospital, adults who battled community-acquired pneumonia were at heightened risk of losing the ability to perform activities and experiencing poor quality of life.

Six months following discharge from the hospital, adults who battled community-acquired pneumonia were at heightened risk of losing the ability to perform activities and experiencing poor quality of life.

Real-world data show that patients switching from Humira to biosimilars Hadlima and Hyrimoz achieve similar outcomes without hospitalization.

Recent research reveals that RSV and shingles vaccines significantly lower dementia risk, highlighting the potential benefits of AS01 adjuvants in vaccination.


LeAnne Kennedy, PharmD, BCOP, CPP, FHOPA, discusses the new outpatient transplant program at Atrium Health Wake Forest Baptist in North Carolina.

Expert explores the expanding role of FDA-approved Prescription Digital Therapeutics (PDTs).

An expert discusses the challenges of diagnosing and treating RSV in older adults and highlights the critical role of prevention.
The letter cited deficiencies previously identified at a third-party manufacturing vendor unrelated to oxylanthanum carbonate (OLC).

Research reveals a link between estrogen plus progestin hormone therapy and increased breast cancer risk in young women, urging personalized treatment approaches.
Vitamin C enhances skin health by promoting epidermal cell growth and regeneration, offering a potential solution for aging and thinning skin.

Discover how Vyepti revolutionizes migraine prevention with rapid efficacy and strong safety, empowering pharmacists to enhance patient care.
Lung transplant recipients were found to be at heightened risk of developing secondary lung infections, including bacterial and fungal infections, due to unique risk factors.

Pharmacy leaders from Cone Health discuss how robotic compounding technologies have improved safety, accuracy, and reliability in hazardous and sterile medication preparation across oncology and acute care settings.
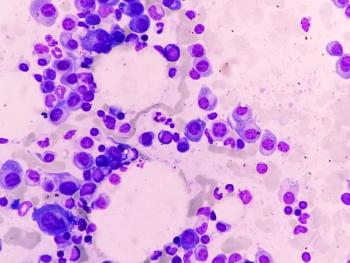
The treatment becomes the first approved BCMAxCD3 bispecific antibody with the potential to achieve biweekly or monthly dosing in patients with relapsed/refractory multiple myeloma.

LeAnne Kennedy, PharmD, BCOP, CPP, FHOPA, discusses the new outpatient transplant program at Atrium Health Wake Forest Baptist in North Carolina.

Manish Agrawal, MD, discusses the implementation of psilocybin-assisted therapy in a community cancer setting, highlighting its potential to provide sustained relief from depression and anxiety and its significant efficacy advantage over selective serotonin reuptake inhibitors for patients with cancer.
The injection is indicated for the treatment of sepsis and other serious infections, enhancing treatment efficiency and patient care.
CARTITUDE-1 trial reveals cilta-cel's potential in achieving long-term remission for relapsed/refractory multiple myeloma, sparking discussions on curative possibilities.
A low-fat vegan diet reduces dietary acid load and promotes weight loss more effectively than a Mediterranean diet.
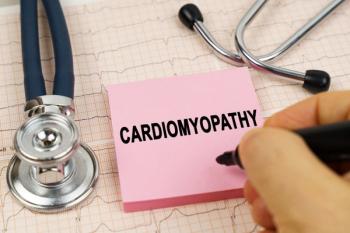
Pharmacists can help oversee the treatment process to ensure patients with transthyretin amyloid cardiomyopathy (ATTR-CM) are adhering to tafamidis.
Obecabtagene autoleucel shows promising efficacy and safety in treating relapsed/refractory B-ALL, benefiting patients of all ages, including older adults.

Simplifying complex information through illustrations has consistently helped patients feel that managing type 1 diabetes is achievable.

The data revealed that only 75% of patients receive HER2-targeting therapies.

Gianni Scappaticci, PharmD, BCOP, outlines a personalized, evidence-based approach to managing steroid-refractory chronic graft-versus-host disease (cGVHD) in a post–haploidentical transplant patient, highlighting the rationale for using ruxolitinib, belumosudil, or their combination based on disease progression and fibrotic features.

Host Craig Beavers sits down with Kazuhiko Kido, PharmD, a clinical associate professor at the West Virginia University School of Pharmacy, to discuss intravenous iron and its use in patients with heart failure.
A direct link between memories and physical changes has not yet been established.
Pharmacists and clinicians collaborate to carefully manage rilonacept therapy.
