
The current acute treatments for migraines have shown efficacy and safety, however, approximately one-third of people do not respond, cannot use these options due to contraindications, or report dissatisfaction in therapy.

The current acute treatments for migraines have shown efficacy and safety, however, approximately one-third of people do not respond, cannot use these options due to contraindications, or report dissatisfaction in therapy.

Study suggests psilocybin may be effective in a much wider population of patients suffering from major depression than previously believed.

Study shows large reductions in visits to essential stores during the pandemic, such as pharmacies, but found essentially no reduction in non-essential visits to parks and other outdoor spots.
Researchers from 2 independent studies have found that SARS-CoV-2 may be more capable of infecting human cells due to the virus’ spike protein recognizing and binding to neuropilin-1.
The FDA granted breakthrough therapy designation to dupilumab in September 2020, for the treatment of patients age 12 years and older with eosinophilic esophagitis.
Angiotensin receptor II blockers are often prescribed to control hypertension or for heart failure, kidney failure, or following a heart attack.

A new blood test can help determine cancer type and whether it will respond to chemotherapy.
Approximately 1 in 3 high school seniors (31.8%) who reported misusing prescription opioids as high school seniors between 1997 and 2000 went on to use heroin by age 35.
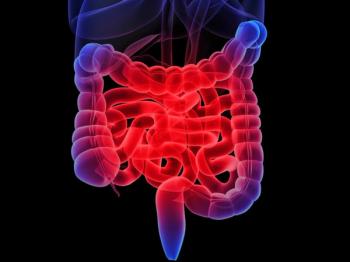
The United States Preventive Services Task Force updates recommends that colorectal cancer screening begin 5 years earlier, at age 45 instead of age 50.

Having a PharmD is an exceptional accomplishment, but it’s what you do with it that will empower you to dominate your career.
Earlier studies have suggested that statins can potentially benefit some forms of cancer and dementia, meaning investigators could design drugs based on these beneficial effects.

The researchers isolated human primary keratinocytes from the skin of patients with non-melanoma skin cancers.
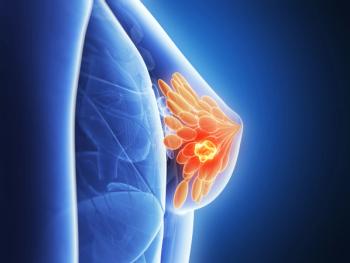
Study to evaluate paclitaxel plus encequidar with dostarlimab +/- carboplatin in patients with breast cancer.

Data show that children with cancer who are experiencing neighborhood poverty and rely on public health insurance have an increased risk of death with stem cell treatment.

Researchers suggest several modifications in the way health professionals manage patients with obesity during the COVID-19 pandemic.
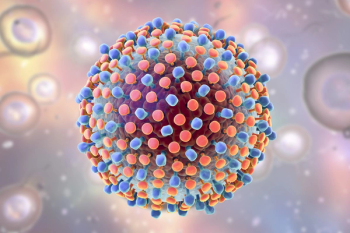
Researchers have identified the role of a protein involved in the progression of hepatitis C virus (HCV) infection that has demonstrated a potential new avenue for treatment.
Simple sugar found in breast milk and dietary supplements may promote myelin repair in patients with multiple sclerosis.

FDA lift clinical hold placed on the phase 1 PSMA-101-001 study of the chimeric antigen receptor T-cell therapy P-PSMA-101 in patients with metastatic castration-resistant prostate cancer.

RG is a male patient aged 45 years who has arrived at the pharmacy to pick up his regular maintenance medications after recently losing 45 pounds in 3 months.

Customers and patients who come to the pharmacy for medication need to feel safe. During COVID-19, that looks a bit different than it does at any other time.

A plethora of new research and CBD products have come on the market in a very short time since the Hemp Farming Act of 2018 removed hemp from Schedule I controlled substances.

Using several sources of data, researchers at Linköping University in Sweden developed a new method to predict flu epidemics.
Dissecting the mechanisms of this immunomodulation could provide important insights into varicella zoster virus pathogenesis and will be important when designing new vaccines and antivirals.

Study suggests COVID-19 cases and deaths in the United States in the fall and winter will far exceed those of spring 2020 unless there is effective implementation of nationally coordinated efforts and preventive strategies.

This week on Pharmacy Times, there are a number of important topics that will be covered and posted throughout the week.

The study used an animal model that was conducted according to the National Advisory Committee for Laboratory Animal Research (NACLAR) guidelines.

Nursing home residents who are transferred to the emergency department potentially face long-term health consequences.
FDA approves the cobas EGFR Mutation Test v2 as a companion diagnostic for EGFR TKIs in the treatment of EGFR-mutated non–small cell lung cancer.

Instagram builds your network in a way that capitalizes on some of the most essential strengths needed as a leader in the profession.

A new study suggests that more than 80% of COVID-19 patients could be vitamin D deficient.