Americans who are fully vaccinated against COVID-19 no longer need to wear masks when outside alone, with members of their household, or when attending small outdoor gatherings.

Americans who are fully vaccinated against COVID-19 no longer need to wear masks when outside alone, with members of their household, or when attending small outdoor gatherings.

According to Michael Sneed, executive vice president of global corporate affairs and chief communication officer for Johnson & Johnson, the health care system has chronically left people of color behind, and the inequities seen throughout the COVID-19 pandemic are only the latest examples.

As the most accessible member of the health care team, the pharmacist can provide patients with cancer with the information and support necessary to get through what could be the most challenging time in their lives.

As new variants of the virus come to rise, it is crucial to continue to address the safety concerns with mass COVID-19 immunizations during the pandemic as well as provide solutions to reduce the exposure and spread of the virus.

Clinicians should consider the impact on subsequent therapies, overall survival, and median time to definitive deterioration with CDK 4/6 inhibitors.
Cynthia Lynch, MD, breast cancer program clinical advisor with Cancer Treatment Centers of America, said oncology pharmacists are vital team members who can help ensure optimal treatment and safety for patients with breast cancer.

Patient and provider organizations urge the Department of Health and Human Services and the Biden administration to enact policies and protections that improve prescription drug affordability for people in the United States.

Sudden disruptions in the supply of street drugs when the COVID-19 pandemic began were associated with an increase in overdose deaths and other harms.
Investigators developed a genetically distinct and non-toxic strain of salmonella called CRC2631 to select and kill cancer cells, which may be able to be tailored to specific patients.

A greater understanding of obesity and its impact on cardiovascular health highlights abdominal obesity, sometimes referred to as visceral adipose tissue (VAT), as a cardiovascular disease risk marker.
Researchers from the University of Pennsylvania School of Medicine have identified additional genetic mutations that strongly predispose otherwise healthy women to peripartum cardiomyopathy (PPCM), a rare condition characterized by weakness of the heart muscle that begins sometime during the final month of pregnancy through 5 months after delivery.

Despite the fact that 98% of the survey respondents said they were aware of PrEP, less than 25% were currently taking it.

The FDA has approved Gemtesa (vibegron; Urovant Sciences) tablets to treat overactive bladder (OAB) with symptoms of urge urinary incontinence (UUI), urgency, and urinary frequency.

For World Immunization Week, Pharmacy Times® interviewed Nurisha Wade, MBA, VP of Global Healthcare Quality & Safety Center of Excellence at US Pharmacopeia (USP), and Farah Towfic, PharmD, MBA, director, CEO of Operations at USP, on USP’s COVID-19 Vaccine Handling Toolkit.

Beattie's Healthmart is known for stellar customer service and its 3 playful, yet protective, dogs.

Pharmacy Times® interviewed Scott J. Knoer, MS, PharmD, FASHP, the EVP and CEO of the American Pharmacists Association, on the Pharmacy and Medically Underserved Area Enhancement Act that was recently introduced in Congress.
The report further showed that drug expenditure in nonfederal hospitals declined 4.6%, while drug spending in home care settings increased 13%, and drug expenditures in nonfederal hospitals spiked in the 3 weeks immediately following the March 8, 2020 lockdown.
The antibiotic fights both resistant enterococcus, streptococcus, and staphylococcus bacteria via a unique mechanism of action, which causes resistance to develop at a very slow pace.

Nocturnal respiratory events, when they either completely or partially block the airways, often cause repeated oxygen desaturations and interrupted sleep in patients with OSA, leading to abnormal nervous system function.
Broadly, the study results showed that if a participant's cognitive ability, physical health, or mental health declined over the course of the 6-month study period, that participant became less cognitively engaged as the tasks increased in difficulty.
Pharmacies cannot prevent shortages, particularly with the complexities of treating COVID-19; however, they can strengthen their ability to prepare their organization to respond to those complexities.
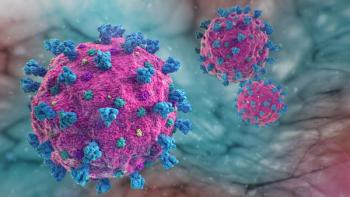
The study found that more than 10% of participants who were previously infected with SARS-CoV-2 (seropositive) became reinfected, compared with new infections in 50% of participants who had not been previously infected (seronegative).
This week on Pharmacy Times, there are a number of important topics that will be covered and posted throughout the week.

A new service connects community pharmacies with health care systems to support the delivery of health care products and services through crowdsourcing.

Face mask usage may help, and pharmacists can provide other education and guidance about safe and proper use of nonprescription medications.
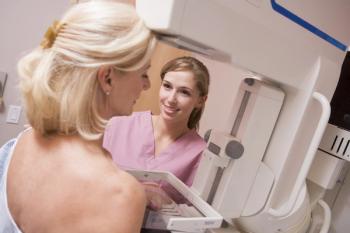
Oncology experts recommend patients schedule overdue screenings, as pandemic restrictions ease.

This is the first systematic literature review on the association of back pain and mortality.
A CDC panel voted to add a warning label to the vaccine regarding rare cases of cerebral venous sinus thrombosis.

Loncastuximab tesirine-lpyl is a CD19-targeted antibody drug conjugate.
Pharmacists and other clinicians share responsibility for relieving symptoms, preventing transmission, and avoiding complications.