
Results of a study by the American Heart Association show a genetic predisposition may lead to an association between sleeplessness and brain bleeding.

Results of a study by the American Heart Association show a genetic predisposition may lead to an association between sleeplessness and brain bleeding.
Rodney Pommier, MD, FACS, professor of surgery, division of surgical oncology, school of medicine, Oregon Health & Science University, discusses his presentation at the 2021 NANETS Virtual Symposium on a prospective study of carcinoid crisis with no perioperative octreotide.
Nicole Ross, MSN, CRNP, AOCNP, nurse practitioner for the department of hematology/oncology at Fox Chase Cancer Center, discusses how communication plays a huge role in treatment of neuroendocrine tumors in large cancer centers.

In a report, the World Health Organization and a partner ask developers, funders, and investigators to accelerate developing a vaccination.
The investigators caution that the quality of the evidence is variable and an optimal formulation or dose remains unknown.
The difference between individuals who were vaccinated and had the infection were higher at different intervals than for those who were unvaccinated.
A new poll shows that this year 64% of individuals between aged 50 and 64 years say they already got or are likely to get their influenza shots up from 54% last year.

Preparation H works by temporarily narrowing the blood vessels in the area, which decreases swelling and discomfort caused by hemorrhoids.
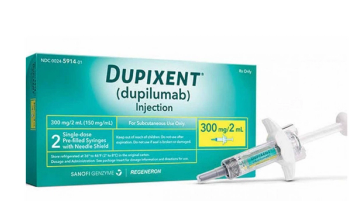
Dupilumab is indicated for the treatment of adult patients with moderate-to-severe atopic dermatitis whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable.
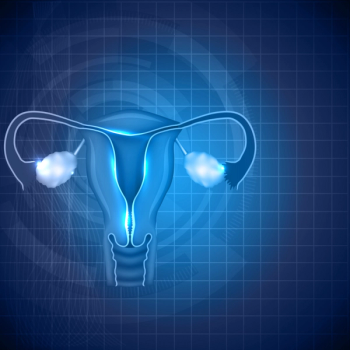
An ectopic pregnancy occurs when a fertilized egg implants and grows outside the main cavity of the uterus.

The FDA also granted Priority Review designation for the toripalimab BLA and set a Prescription Drug User Fee Act action date for April 2022; however, the FDA is not currently planning to hold an advisory committee meeting to discuss the application.

In addition to the prevention of disease in children, COVID-19 vaccination can also limit disruptions in school attendance so that children can participate safely in school and other activities.
Kaushal Nanavati, MD, assistant dean of wellness and director of integrative medicine and survivorship at the Upstate Cancer Center discusses why integrative treatment can help physicians personalize treatments for individuals with NETs and why it is beneficial to improve the quality of life for them.

Study shows that about half the pediatric cases were symptomatic compared with 88% for individuals aged 18 years and older.

The AHA emphasizes the importance of looking at a total dietary pattern, including amounts, balance, combinations, and variety of beverages and food consumer regularly.

Agency's interpretation may significantly affect how health systems and hospitals provide pharmaceuticals to patients.
Mei Ka Fong, PharmD, BCOP, said there are multiple treatment approaches for patients with NETs.

The CVS Health 2021 Health Care Insights Study found significant differences in how patients of various ages and genders approach care.
Nicole Ross, MSN, CRNP, AOCNP, nurse practitioner for the department of hematology/oncology at Fox Chase Cancer Center, discusses what kind of support services at large cancer centers can help individuals with neuroendocrine tumors with their treatment.
The study also collected patient-reported outcomes in routine clinical practice to understand the impact of treatment on health-related quality of life.
TNBC is the most aggressive and lethal form of the disease.
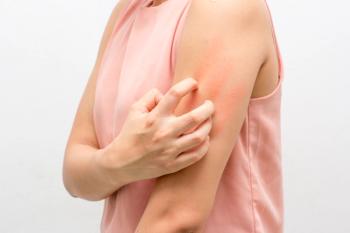
Tralokinumab is a high-affinity human monoclonal antibody that binds to and inhibits interleukin-13, a key driver of atopic dermatitis symptoms.
Kaushal Nanavati, MD, assistant dean of wellness and director of integrative medicine and survivorship at the Upstate Cancer Center, discusses how integrative medicine can help treatment of neuroendocrine tumors in combination with other treatments.

Pharmacists and technicians have an opportunity to prove their value as immunizers now that the Pfizer-BioNTech COVID-19 vaccine has been authorized for use in children ages 5 to 11.

Ten quiz questions to assess your knowledge on common symptoms and treatments for migraine headaches.

This is the first large-scale transdiagnostic study of objectively measured sleep and mental health.

Having community pharmacists to influence outcomes associated with social determinants of health in the US population is vital to improving overall patient outcomes.

Women leadership roles and mentorship is extremely important in the pharmacy field, and this week's episode features Dr. Andrea Collaro, Walgreens Boots Alliance, Senior Director, Brand Management/Product Development, Owned Brands Health & Wellness, who goes into more specifics.
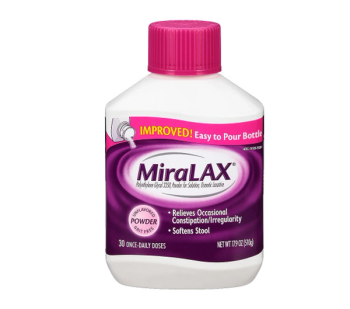
MiraLAX is used as a laxative to treat occasional constipation or irregular bowel movements.
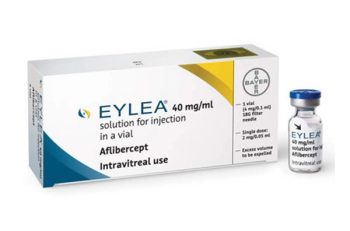
Aflibercept is indicated for the treatment of patients with neovascular (wet) age-related macular degeneration, macular edema following retinal vein occlusion, diabetic macular edema, and diabetic retinopathy.