Top news of the day from across the health care landscape.

Top news of the day from across the health care landscape.

Study finds that glecaprevir/pibrentasvir pan-genotypic efficacy for hepatitis C virus is achieved regardless of whether patients are concurrently receiving gastric acid-reducing drugs.

The research centers on CD8+ T cells, which can recognize and attack the influenza virus.
The new formulation will provide patients with a faster alternative for management of suspected myocardial infarction.

Indivior’s once-monthly schizophrenia treatment risperidone (Perseris) extended-release injectable suspension has launched in the United States.









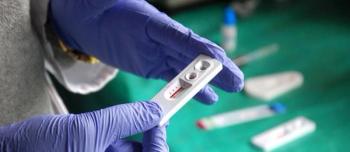
According to the CDC, major interventions are in need among at-risk populations in order to reach a goal of 90% decrease of HIV infections in 10 years.

The CDC has declared immunizations one of the greatest health achievements of the 20th century, but vaccination rates among adults remain suboptimal.

Trastuzumab and hyaluronidase-oysk (Herceptin Hylecta, Genentech) is indicated for subcutaneous administration in certain patients with HER2-positive early and metastatic breast cancer.

Trastuzumab and hyaluronidase-oysk (Herceptin Hylecta, Genentech) is indicated for subcutaneous administration in certain patients with HER2-positive early and metastatic breast cancer.